 Li K, Song X, Ning P, Yi H, Tang X, Wang C (2015) Energy utilization of yellow phosphorus tail gas: simultaneous catalytic hydrolysis of carbonyl sulfide and carbon disulfide at low temperature. Ning P, Li K, Yi H, Tang X, Peng J, He D, Zhao S (2012) Simultaneous catalytic hydrolysis of carbonyl sulfide and carbon disulfide over modified microwave coal-based active carbon catalysts at low temperature. 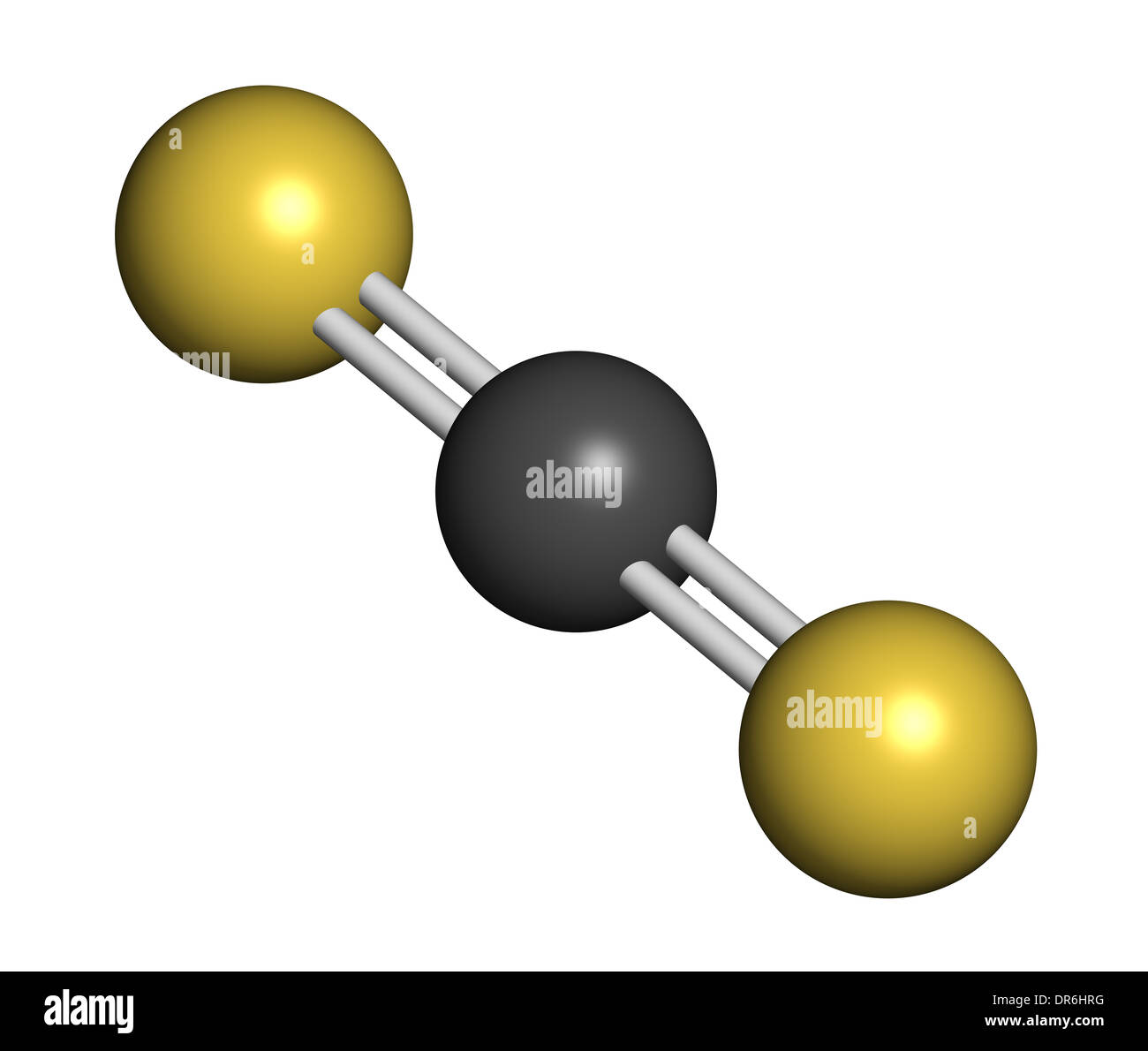
Li K, Tang X, Yi H, Ning P, Kang D, Wang C (2012) Low-temperature catalytic oxidation of NO over Mn–Co–Ce–Ox catalyst. Ling L, Zhang R, Han P, Wang B (2012) A theoretical study on the hydrolysis mechanism of carbon disulfide. 
Saju A, Mondal A, Chattopadhyay T, Kolliyedath G, Kundu S (2020) H 2S generation from CS 2 hydrolysis at a dinuclear zinc (II) site. Liu N, Ning P, Sun X, Wang C, Song X, Wang F, Li K (2021) Simultaneous catalytic hydrolysis of HCN, COS and CS 2 over metal-modified microwave coal-based activated carbon. Li K, Liu G, Gao T, Lu F, Tang L, Liu S, Ning P (2016) Surface modification of Fe/MCSAC catalysts with coaxial cylinder dielectric barrier discharge plasma for low-temperature catalytic hydrolysis of CS 2. 
Song X, Chen X, Sun L, Li K, Sun X, Wang C, Ning P (2020) Synergistic effect of Fe 2O 3 and CuO on simultaneous catalytic hydrolysis of COS and CS 2: experimental and theoretical studies. ĭanten Y, Cabaço MI, Coutinho JAP, Pinaud N, Besnard M (2016) DFT study of the reaction mechanisms of carbon dioxide and its isoelectronic molecules CS 2 and OCS dissolved in pyrrolidinium and imidazolium acetate ionic liquids. Zeng Z, Dlugogorski BZ, Oluwoye I, Altarawneh M (2019) Co-oxidation of methane (CH 4) and carbon disulfide (CS 2). Song X, Ning P, Wang C, Li K, Tang L, Sun X, Ruan H (2017) Research on the low temperature catalytic hydrolysis of COS and CS 2 over walnut shell biochar modified by Fe–Cu mixed metal oxides and basic functional groups. Iglesias-Sigüenza FJ (2008) Carbon disulfide (CS 2). Wang F, Chen H, Sun X, Wang C, Ma Y, Song X, Li K, Ning P, He H (2021) Single atom Fe in favor of carbon disulfide (CS 2) adsorption and thus the removal efficiency. J Quant Spectrosc Radiat Transfer 243:106836. Zhang Y, Wang Y, Liu Y, Ai X, Zhang Z, Li J (2020) Measurement of the absorption cross sections of unstable CS based on the dynamic equilibrium of CS 2 and CS in the photolysis of CS 2. Ĭhalansonnet M, Carreres-Pons M, Venet T, Thomas A, Merlen L, Boucard S, Pouyatos B (2020) Effects of co-exposure to CS 2 and noise on hearing and balance in rats: continuous versus intermittent CS 2 exposures. Ĭhen X, Li J, Jia C (2019) Thermodynamic properties of gaseous carbon disulfide. Just toxic or also bioregulatory and/or therapeutic? Chem Soc Rev 46:21–39. ĭeMartino AW, Zigler DF, Fukuto JM, Ford PC (2017) Carbon disulfide. Zhang X, Ponce V, Galvez-Aranda DE, Zhou G, Zhou H, Seminario JM (2021) CS 2 removal from C 5 distillates by reactive molecular dynamics simulations. Yue X, Wang H, Kong J, Li B, Yang J, Li Q, Zhang J (2020) A novel and green sulfur fertilizer from CS 2 to promote reproductive growth of plants. Liu N, Song X, Wang C, Li K, Ning P, Sun X, Wang F, Ma Y (2020) Surface characterization study of corn-straw biochar catalysts for the simultaneous removal of HCN, COS, and CS 2. Zeng Z, Dlugogorski BZ, Oluwoye I, Altarawneh M (2019) Combustion chemistry of carbon disulphide (CS 2). Wang W, Fan L, Wang G (2017) Carbon disulfide (CS 2) adsorption and dissociation on the Cu(100) surface: a quantum chemical study. Our results might contrite to provide a novel method to study the catalytic hydrolysis of CS 2. During the hydrolysis of CS 2, the five reaction paths are parallel and competitive. It was sought out that the hydrolysis of CS 2 was an exothermic reaction, and the increment in temperature was adverse to the reaction. Besides, to further clarify which reaction path is better, the hydrolysis kinetics of CS 2 was analyzed. Apart from this, to further explore the reaction of CS 2 hydrolysis, the natural bond orbital (NBO) analysis of the transition states was carried out. 
Similarly, the order of the barriers for the three paths in the second reaction is C = S path < C = S path and C = O path < C = O path. In the reaction’s first step, the rate-determining step in both the single C = S path and the double C = S path possesses a higher barrier of 199.9 kJ/mol however, the 127.9 kJ/mol barrier in the double C = S path is lower than the 142.8 kJ/mol in the single C = S path. Hydrogen migration is crucial to the mechanism of CS 2 hydrolysis. Through optimizing the structure of reactants, intermediates, transition states, and products, it can be seen that the first step of CS 2 is that CS 2 reacts with H 2O first to form COS intermediate and the second step is COS intermediate reacts with H 2O to form H 2S and CO 2. Density functional theory (DFT) is used to look into the two-step hydrolysis mechanism of CS 2. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
